
Electronegativity Effect
The resonance position of protons bonded to carbon is shifted down field by electronegative elements also bonded to the carbon. The electronegative element withdraws electron density from the carbon and its directly bonded protons which diminishes the magnitude of the Be field. Since the Be field shields the protons from the Bo field, a smaller (in magnitude) Be field requires a lower Bo field to achieve resonance, hence a downfield shift. The higher the electronegativity of the directly bonded atom, the larger the downfield shift. (see Equation 2 and Figure 4). The higher the electronegativity of the directly bonded atom, the larger the down field shift.

![]() -Bond Effect
-Bond Effect
Alkenes and aromatic compounds bearing ![]() -bonds will spend a portion of their time oriented with the plane defined by their sigma bonds perpendicular to the direction of the Bo field. In this orientation, the
-bonds will spend a portion of their time oriented with the plane defined by their sigma bonds perpendicular to the direction of the Bo field. In this orientation, the ![]() -electrons will circulate to produce a Be field that opposes the Bo field above and below the
-electrons will circulate to produce a Be field that opposes the Bo field above and below the ![]() -bond but adds to the Bo field in the vicinity of the hydrogen nuclei as shown in Figure 7. Since the Be field from the
-bond but adds to the Bo field in the vicinity of the hydrogen nuclei as shown in Figure 7. Since the Be field from the ![]() -electrons is augmenting the applied field, a lower applied field at fixed B1 frequency is required to achieve resonance. Because this mechanism causes alkene and aromatic hydrogen nuclei to resonate down field, they are also said to be deshielded by the
-electrons is augmenting the applied field, a lower applied field at fixed B1 frequency is required to achieve resonance. Because this mechanism causes alkene and aromatic hydrogen nuclei to resonate down field, they are also said to be deshielded by the ![]() -electrons but in reality the contribution to the total Be field from the
-electrons but in reality the contribution to the total Be field from the ![]() -electrons is just opposite in sign to the contribution from the other electrons. In contrast, the major effect from
-electrons is just opposite in sign to the contribution from the other electrons. In contrast, the major effect from ![]() -electrons in the terminal alkyne occurs when the molecular axis lies along the direction of the Bo field. In this orientation the
-electrons in the terminal alkyne occurs when the molecular axis lies along the direction of the Bo field. In this orientation the ![]() -electrons circulate about the girth of the molecule involving both
-electrons circulate about the girth of the molecule involving both ![]() -bonds to produce a contribution to the Be field which opposes the Bo field in the vicinity of the terminal proton (see Figure 7, below). Hence, the terminal alkyne proton resonates upfield (
-bonds to produce a contribution to the Be field which opposes the Bo field in the vicinity of the terminal proton (see Figure 7, below). Hence, the terminal alkyne proton resonates upfield (![]() ~2.5 ppm) from the alkene (
~2.5 ppm) from the alkene (![]() ~5.5 ppm) and aromatic protons (
~5.5 ppm) and aromatic protons (![]() ~7 ppm). The effect from the
~7 ppm). The effect from the ![]() -electrons is an anisotropic effect because its magnitude and direction depend upon a particular orientation of the molecules with respect to the direction of the Bo field. An isotropic effect is independent of orientation.
-electrons is an anisotropic effect because its magnitude and direction depend upon a particular orientation of the molecules with respect to the direction of the Bo field. An isotropic effect is independent of orientation.

The chemical shifts of some hydrogen nuclei (protons) in various magnetic environments are shown in Figure 8. A general rule to remember about chemical shifts of protons on saturated carbon is that the methylene proton (CH2) chemical shift will appear approximately 0.2-0.4 ppm downfield from the corresponding methyl proton chemical shift, and the methine proton (CH) chemical shift will appear approximately 0.2-0.4 ppm downfield from the corresponding methylene chemical shift: ![]() CH>CH2>CH3. This is because carbon is slightly more electronegative than hydrogen. Electronegativity and
CH>CH2>CH3. This is because carbon is slightly more electronegative than hydrogen. Electronegativity and ![]() -bond effects are additive; additivity is readily apparent in the chemical shift of the aldehyde proton (
-bond effects are additive; additivity is readily apparent in the chemical shift of the aldehyde proton (![]() = 10) and of the methoxy of a methyl ester (
= 10) and of the methoxy of a methyl ester (![]() = 4) relative to the methoxy of an ether (
= 4) relative to the methoxy of an ether (![]() = 3.5).
= 3.5).
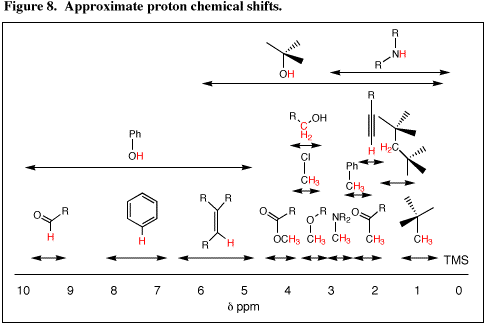
From Figure 8 we quickly see that the position of absorption is suggestive of the presence of functional groups. A peak between ![]() 9 and 10 indicates an aldehyde, peaks in the region
9 and 10 indicates an aldehyde, peaks in the region ![]() 7 to 8 indicate an aromatic ring, peaks in the region
7 to 8 indicate an aromatic ring, peaks in the region ![]() 5 to 6 indicates an alkene, peaks between
5 to 6 indicates an alkene, peaks between ![]() 3.5 to 4.5 indicate an ester, etc. Note that protons on the heteroatoms O and N show a wide range of chemical shifts which are concentration, temperature, solvent, and pH dependent. These protons are involved in hydrogen bonding and exchange with each other and with water (usually present as an impurity) by an acid-base mechanism through hydrogen bonding. Variable hydrogen bonding and exchange give rise to the variable chemical shift.
3.5 to 4.5 indicate an ester, etc. Note that protons on the heteroatoms O and N show a wide range of chemical shifts which are concentration, temperature, solvent, and pH dependent. These protons are involved in hydrogen bonding and exchange with each other and with water (usually present as an impurity) by an acid-base mechanism through hydrogen bonding. Variable hydrogen bonding and exchange give rise to the variable chemical shift.
The following is a summary of electronegativity and ![]() -bond effects on chemical shift. For your convenience, this and other reference information is included in the Reference section of this tutorial.
-bond effects on chemical shift. For your convenience, this and other reference information is included in the Reference section of this tutorial.

next section: Chemical Shift - Carbon
![]()
Copyright information: Original content © University of Colorado, Boulder, Chemistry and Biochemistry Department, 2011. The information on these pages is available for academic use without restriction.